What is TMS?
NeuroStim TMS Therapy for Depression
What is TMS? Transcranial Magnetic Stimulation (TMS) is a technological breakthrough in the treatment of Major Depressive Disorder (MDD), Treatment-Resistant Depression (TRD), Post Traumatic Stress Disorder (PTSD), Obsessive-Compulsive Disorder (OCD), Anxiety, and other mood disorders. It is an FDA-approved, non-invasive treatment that has been recognized by the American Psychiatric Association in its ‘Best Practices Guidelines’ as an effective treatment for major depression.
NeuroStim TMS is a safe and effective outpatient treatment for individuals diagnosed with Major Depressive Disorder (MDD) or similar disorders who have not experienced satisfactory improvement from antidepressant medication or other treatments.
Watch the short video of Jonathan Downar, MD, PhD explaining NeuroStim TMS:
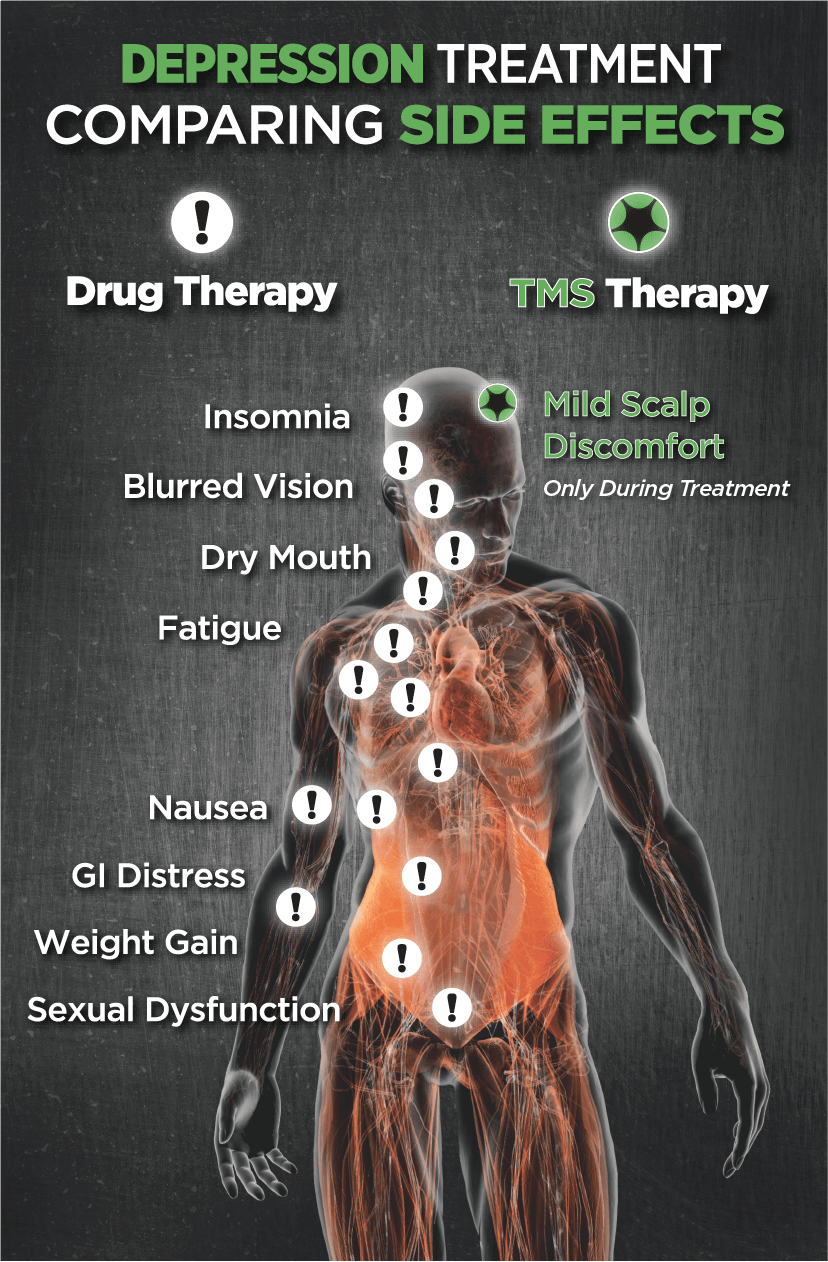
TMS Therapy is Not Invasive
TMS is non-invasive, meaning that it does not involve any anesthesia or sedation, as the patient remains awake and alert during the treatment. It is also non-systemic, meaning that it does not circulate in the bloodstream throughout the body. Side effects frequently experienced from antidepressant medications (i.e. weight gain, sexual dysfunction, tiredness, etc) do not occur with TMS therapy. There is no recovery time either, so patients can drive home afterwards and resume their daily activities.
TMS Therapy is Effective
In FDA clinical trials, 2 out of every 3 patients reported remission or a significant reduction in depression symptoms after completion of TMS Therapy. Subsequent studies have consistently shown at least 3 out of every 4 patients positively responding to NeuroStim TMS Therapy.
NeuroStim TMS is a leading national TMS Provider with sixteen (16) clinic locations conveniently located across Washington, Arizona, and Minnesota. NeuroStim TMS has performed over 250,000 successful treatments to date.
Listen to Lindsey’s story of beating her depression with NeuroStim TMS.
Listen to Lindsey’s story of beating her depression with NeuroStim TMS.